Summit Therapeutics (SMMT)·Q4 2025 Earnings Summary
Summit Therapeutics Ends 2025 With $713M Cash, Eyes Q2 Catalyst for Keytruda Challenger
February 23, 2026 · by Fintool AI Agent
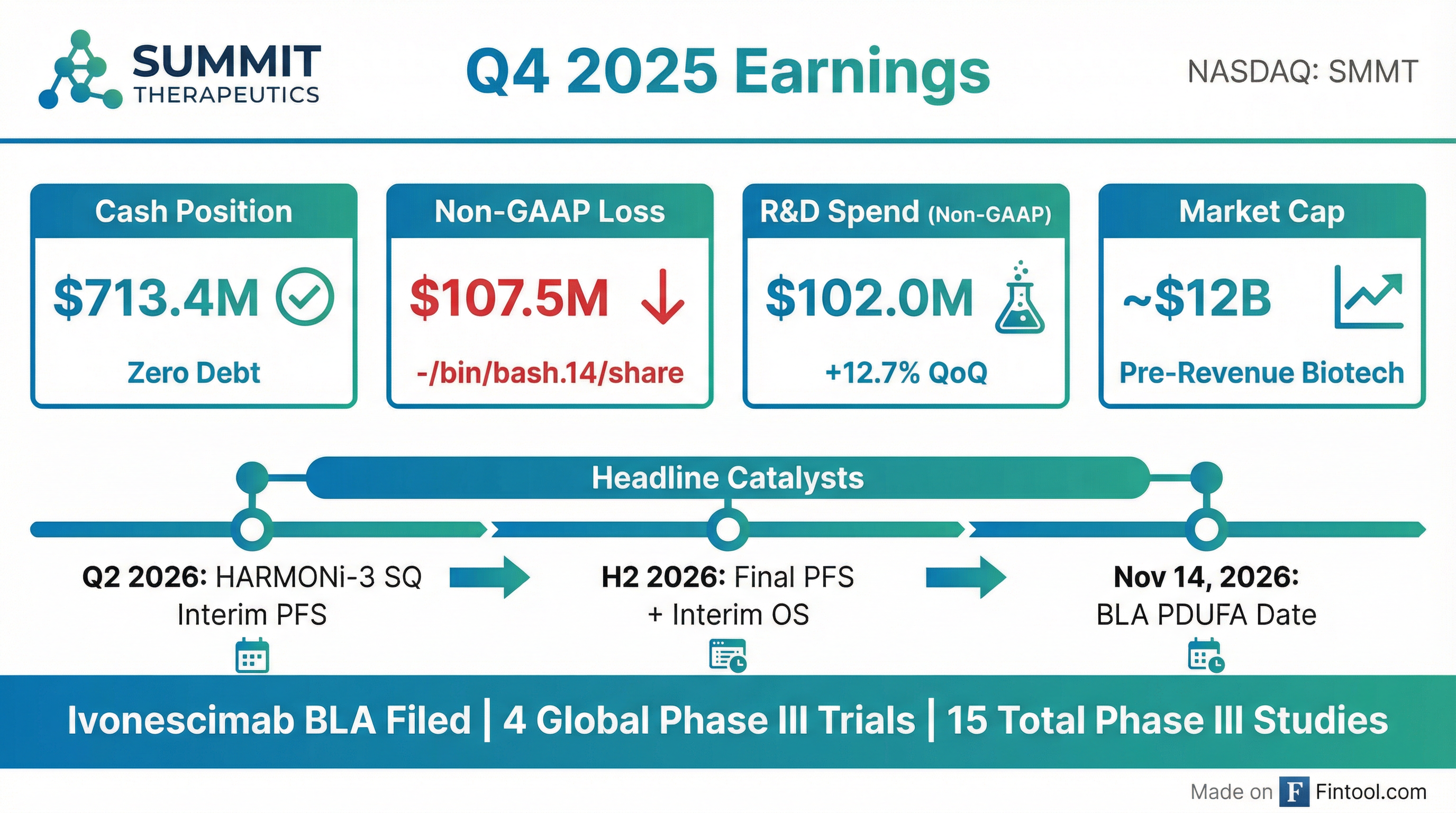
Summit Therapeutics reported Q4 2025 results on February 23, 2026, highlighting a strengthened balance sheet of $713.4 million in cash with zero debt and a packed catalyst calendar for 2026. The pre-revenue biotech is advancing ivonescimab, a first-in-class PD-1/VEGF bispecific antibody that could challenge Merck's Keytruda in front-line lung cancer. With the FDA BLA accepted (PDUFA Nov 14, 2026) and pivotal HARMONi-3 data expected starting Q2 2026, the year ahead is critical for Summit's thesis as a potential oncology platform company.
Key call highlights: Patient screening completed for HARMONi-3 squamous cohort with last patient randomization expected "in the next couple of weeks" ; manufacturing process successfully transferred and validated at a US-based manufacturer ; over 60,000 patients have now received ivonescimab commercially in China across two approved indications .
What Are the Key Financial Results?
Summit is a clinical-stage biotech with no revenue. For pre-revenue biotechs, the key financial metrics are cash position and operating expense trajectory.
The large GAAP/Non-GAAP gap is driven by stock-based compensation of $111.7 million in Q4 ($732.4M for FY 2025), primarily related to modifications of performance-based stock option awards in Q2 2025.
Full Year 2025 Summary:
- Non-GAAP R&D: $319.2M (vs. $134.8M in 2024) — reflecting expansion of HARMONi-3, HARMONi-7, and initiation of new trials
- Non-GAAP G&A: $42.8M (vs. $25.2M in 2024), with quarterly run rate of ~$10-11M — building infrastructure to support commercial readiness
- Cash used in operations: $322.9M (vs. $142.1M in 2024)
- Cash from financing: $617.5M raised during 2025
What Changed From Last Quarter?
Several important updates emerged since the Q3 2025 report:
BLA Filing & FDA Acceptance: In Q4 2025, Summit submitted its Biologics License Application for ivonescimab based on HARMONi trial results. In January 2026, the FDA accepted the filing with a PDUFA goal action date of November 14, 2026.
HARMONi-3 Protocol Update: The statistical plan was amended to include an interim PFS analysis for the squamous cohort. Patient screening for the squamous cohort completed ahead of plan, with the last patient to be randomized "in the next couple of weeks." Management credited the team for "accelerating the enrollment of 600 squamous patients ahead of our planned timelines."
New Collaborations Advancing:
- Revolution Medicines: First patient dosed in Q1 2026 evaluating ivonescimab + RAS(ON) inhibitors
- GSK: Clinical trials with novel B7-H3 (risvutatug rezetecan) expected to start mid-2026
US Manufacturing Ready: Summit successfully transferred and validated the production process of ivonescimab to a US-based manufacturer — a critical step for commercial readiness.
ILLUMINE Phase III Announced: GORTEC will conduct a Phase III study in 1L PD-L1 positive recurrent/metastatic HNSCC (head and neck squamous cell carcinoma) with first patient expected early Q2 2026.
What Are the Upcoming Catalysts?
2026 is shaping up to be a transformational year for Summit with multiple data readouts and regulatory milestones:
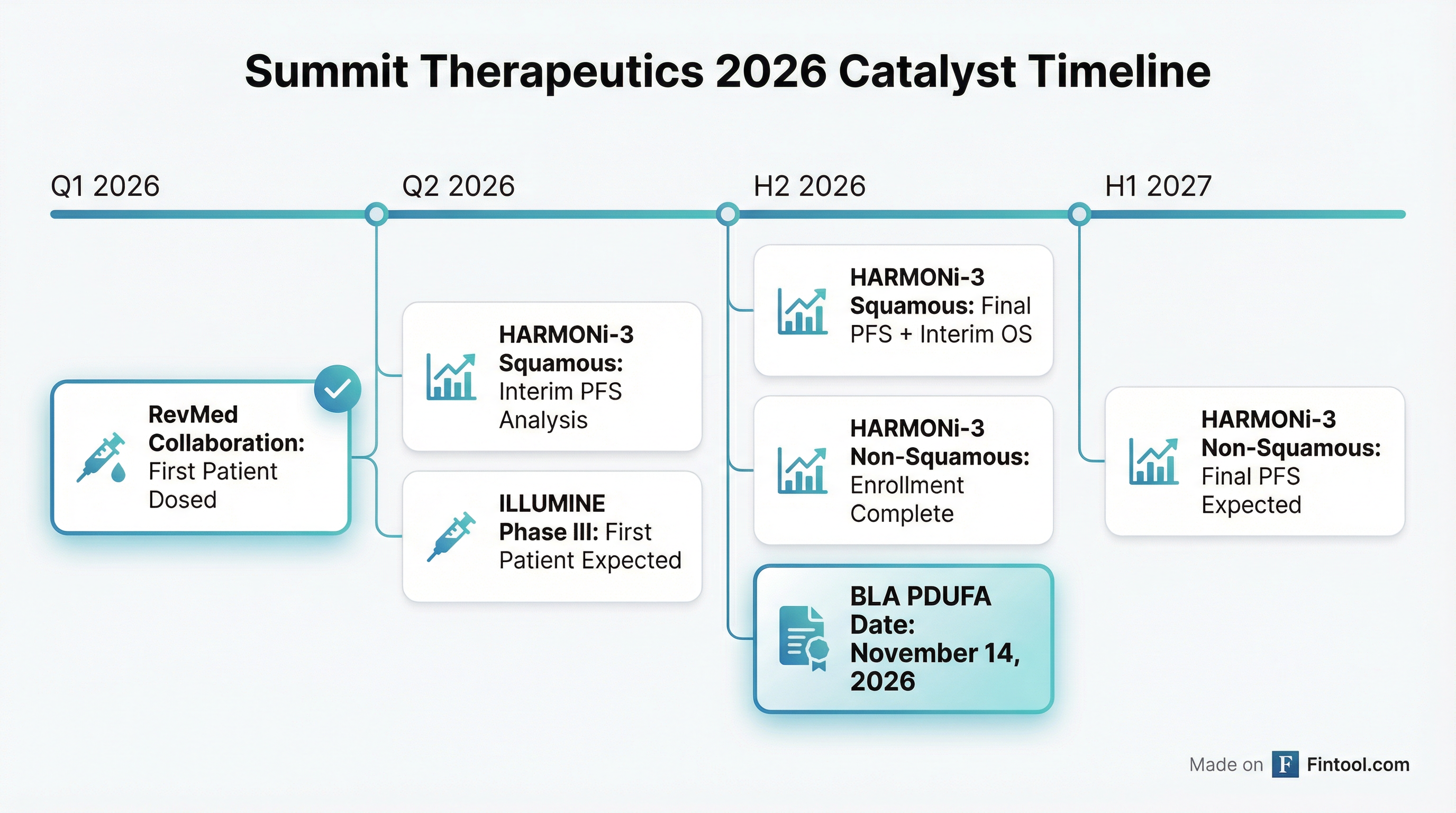
The Q2 2026 interim PFS analysis for HARMONi-3 squamous is the most anticipated near-term catalyst. Note that overall survival is expected to be immature at the time of this interim analysis.
How Did the Stock React?
SMMT closed at $16.09 on February 23, 2026, up +0.94% on earnings day. The stock showed muted reaction as results were largely in-line with expectations and near-term catalysts remain months away.
Key context:
- 52-week range: $13.83 - $36.91
- Market cap: ~$12 billion
- Stock down ~56% from 52-week high hit after HARMONi-6 data at ESMO 2025
The stock saw significant volatility in 2025, peaking after positive HARMONi-6 Phase III data was presented at ESMO in September 2025, which showed ivonescimab + chemo significantly outperformed tislelizumab + chemo in squamous NSCLC with a hazard ratio of 0.60 (p<0.0001).
What Did Management Say in Q&A?
The Q&A session provided critical color on regulatory strategy and clinical confidence:
On adding interim PFS analysis to HARMONi-3:
"We decided to amend the protocol for the HARMONi-3 study by including an interim analysis for the PFS primary endpoint... It's really about accelerating the timelines with respect to the data based on two interim readouts from our partners at Akeso in studies in lung cancer." — Dave Gancarz, CBSO
On confidence in PFS translating to OS:
"Of the four [Phase III studies] that have read out, three of those studies have shown some data towards OS. All of them have shown a hazard ratio of less than 0.8, which when we speak to KOLs, we speak to physicians globally, that's kind of the generally agreed upon threshold for clinical meaningfulness." — Dave Gancarz
On commercial readiness:
"EGFR is the smallest one, right, to initiate. We don't have to put a lot of expenditure, and the most of the expenditure will come, right, when we hire our sales teams over there... There are a lot of synergies, right? If you see our EGFR, and squamous and non-squamous all are coming from the non-small cell lung cancer." — Manmeet Soni, COO/CFO
On FDA interactions:
"We have interactions with the agency. We don't necessarily disclose, meeting by meeting discussions... We have, we do have interactions with the agency, both for this study as well as other current studies and then potential future studies." — Dave Gancarz
On OS expectations (managing investor expectations):
"Once we get the data, the most important thing is trying to provide this agent to patients as soon as possible. That requires a regulatory interaction, right? As a courtesy to them, we need to demonstrate to them first." — Allen Yang, Chief of R&D Strategy
Why Does This Matter for Investors?
The Bull Case:
- Ivonescimab is a potential best-in-class PD-1/VEGF bispecific with four positive Phase III readouts to date — "the only phase III readouts that we have seen in the PD-1/VEGF bispecific class to date"
- Addresses a $100B+ total addressable market combining checkpoint inhibitors and anti-VEGF therapies (per TD Cowen estimates)
- Strong balance sheet ($713M cash, no debt) provides runway through multiple data readouts
- BLA already accepted — first potential approval Nov 2026
- Broad platform potential with 15 Phase III trials and 142 total clinical trials on ClinicalTrials.gov; over 4,000 patients enrolled in Summit/Akeso-sponsored trials
The Bear Case:
- Still pre-revenue with accelerating cash burn (~$107M/quarter non-GAAP)
- HARMONi-3 must succeed vs. pembrolizumab (Keytruda) — the gold standard
- FDA previously indicated statistically significant OS required for HARMONi approval (interim OS trended positive but not statistically significant)
- Competition from other PD-1/VEGF combinations and ADCs
- Stock down significantly from highs despite positive data
What Changed in 2025: Summit executed one of the fastest Phase III ramp-ups in biotech history, going from a single study to four global Phase III trials while raising >$600M. The company is now well-funded for its catalyst-rich 2026.
What Should Investors Watch?
-
Q2 2026 HARMONi-3 Squamous Interim PFS — The most important near-term catalyst. Strong results could validate the platform and rerate the stock.
-
November 2026 PDUFA — First potential US approval for ivonescimab, though in a smaller post-TKI EGFRm indication.
-
Cash Runway — At ~$107M/quarter non-GAAP burn and $713M cash, Summit has approximately 6-7 quarters of runway. Additional financing likely needed in 2026-2027.
-
Overall Survival Maturation — While HARMONi-6 showed strong PFS, OS was not mature. Maturing OS data across studies will be critical for full regulatory approval in larger indications.
Chairman's Closing Remarks
Bob Duggan, Chairman and Co-CEO, closed the call with notable comments:
"Not only is David correct in saying that we have a tremendous respect for the FDA, it is probably America's most respected agency around the world for its integrity, the due diligence of its work, putting patients first... If there's any China lookalike, a Regeneron, it's Akeso, just a fabulous company with great engineers, great scientists, and we're pleased that they are the source of the bispecific tetravalent back in 2013."
Related Resources
- Summit Therapeutics Company Profile
- Q4 2025 Earnings Transcript
- Q3 2025 Earnings Transcript
- Latest SEC Filings
Summit Therapeutics (NASDAQ: SMMT) is a clinical-stage biopharmaceutical company focused on developing ivonescimab (SMT112), a potential first-in-class bispecific antibody targeting PD-1 and VEGF for solid tumors. The company is headquartered in Miami, Florida.